Online Pet Vet Pharmacy: Best Deals Up to 40% Off + FREE Shipping on Orders €100+


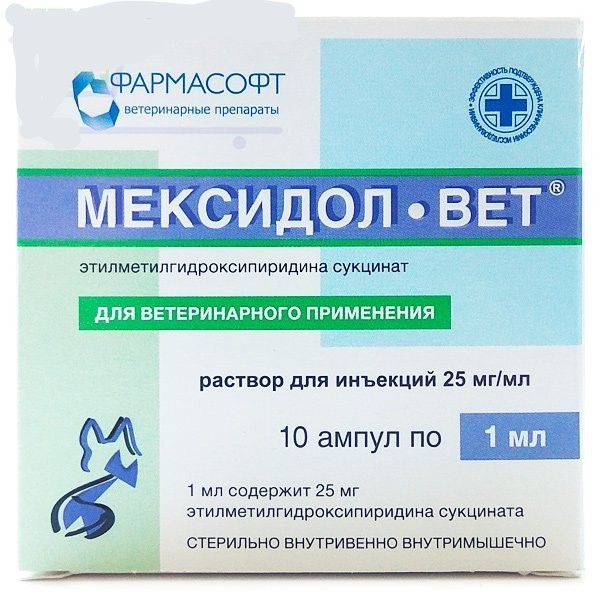
Mexidolvet for dogs and cats – 10 amp for injection
39.00 € Original price was: 39.00 €.32.67 €Current price is: 32.67 €.
Prescribed to dogs and cats for prophylactic and therapeutic purposes independently and in complex therapy for acute and chronic cardiovascular and cardiopulmonary insufficiency, acute pyoinflammatory processes in the abdominal cavity (acute necrotizing pancreatitis, peritonitis), in order to prevent anesthetic and post-narcotic complications , in the post-traumatic and postoperative periods, including with traumatic brain injuries, with pathology of the skin (dermatitis, eczema, trophic ulcers and wounds), to improve the condition of the skin and hair, in veterinary geriatrics (caring for old animals) with age dysfunctions of the animal’s body (sluggish chronic diseases, neurodermatitis, dermatitis associated with impaired peripheral circulation).
| Animals | Cats, Dogs |
|---|---|
| Brands | Farmasoft |
| Form | Suspension/Emulsion/Syrup |
Out of stock
Mexidolvet for dogs and cats – 10 amp for injection
COMPOSITION AND FORM OF ISSUE
1 ml of Mexidol-Vet solution for injection contains 25 mg or 50 mg of mexidol (ethylmethylhydroxypyridine succinate) as an active ingredient, as well as water for injection. In appearance it is a colorless or slightly yellowish transparent liquid. Produced in the form of 2.5% and 5% sterile solution, packaged in glass ampoules of 1 ml and 2 ml, respectively. Packed in 5 ampoules in blister packs made of polyvinyl chloride film, which are put in 2 pieces in cardboard boxes.
PHARMACOLOGICAL PROPERTIES
Mexidol-Vet has pronounced antioxidant, antihypoxic and membrane-protective properties. Increases the resistance of the animal’s body to the effects of various damaging factors to oxygen-dependent pathological conditions (shock, hypoxia, ischemia). Improves microcirculation and rheological properties of blood, stabilizes the membrane structures of platelets and erythrocytes during hemolysis, has a hypolipidemic effect, reduces total cholesterol and low density lipoproteins. Mexidol-Vet inhibits lipid peroxidation, increases the activity of superoxide dismutase, improves synaptic transmission and transport of mediators. The mechanism of action of the drug lies in the ability to cause an increase in the compensatory activation of aerobic glycolysis and to reduce the degree of inhibition of oxidative processes in the Krebs cycle under hypoxic conditions, with an increase in the content of ATP and creatine phosphate, to activate the energy synthesizing function of mitochondria. When administered parenterally, Mexidol-Vet quickly enters the blood, organs and tissues of the animal. It is excreted from the body of the animal in the urine, mainly in the glucuronoconjugated form. Mexidol-Vet solution for injection, according to the degree of effect on the body of warm-blooded animals, refers to moderately hazardous substances, does not possess local irritating, cumulative, embryotoxic and teratogenic properties. When administered parenterally, Mexidol-Vet quickly enters the blood, organs and tissues of the animal. It is excreted from the body of the animal in the urine, mainly in the glucuronoconjugated form. Mexidol-Vet solution for injection, according to the degree of effect on the body of warm-blooded animals, refers to moderately hazardous substances, does not possess local irritating, cumulative, embryotoxic and teratogenic properties. When administered parenterally, Meksidol-Vet quickly enters the blood, organs and tissues of the animal. It is excreted from the body of the animal in the urine, mainly in the glucuronoconjugated form. Meksidol-Vet solution for injection, according to the degree of effect on the body of warm-blooded animals, refers to moderately hazardous substances, does not possess local irritating, cumulative, embryotoxic and teratogenic properties.
INDICATIONS
Prescribed to dogs and cats for prophylactic and therapeutic purposes independently and in complex therapy for acute and chronic cardiovascular and cardiopulmonary insufficiency, acute pyoinflammatory processes in the abdominal cavity (acute necrotizing pancreatitis, peritonitis), in order to prevent anesthetic and post-narcotic complications , in the post-traumatic and postoperative periods, including with traumatic brain injuries, with pathology of the skin (dermatitis, eczema, trophic ulcers and wounds), to improve the condition of the skin and hair, in veterinary geriatrics (caring for old animals) with age dysfunctions of the animal’s body (sluggish chronic diseases, neurodermatitis, dermatitis associated with impaired peripheral circulation).
DOSES AND METHOD OF APPLICATION
Mexidol-Vet is administered to animals intramuscularly or intravenously (drip – at a rate of 60 drops per minute, jet – within 5 – 7 minutes). When administered intravenously, the drug is diluted with water for injection or saline. The therapeutic dose of the drug depends on the severity of the clinical course of the disease. In the acute period of the disease, the drug is used starting with a dose of 10-15 mg per 1 kg of animal weight, then the dose is reduced to 5 mg per 1 kg of animal weight. The course of treatment is 5-15 days. The daily dose of Meksidol-Vet solution for injection is administered in two doses. In order to prevent anesthetic and post-anesthetic complications, the drug is used 2 – 3 days before surgery, in the postoperative period – within 2 – 3 days after surgery, 2 times a day. With prolonged anesthesia and a severe clinical condition of the animal, the drug is administered during the operation in fractional doses of 20 mg per 1 kg of animal weight. As a therapeutic and prophylactic agent in veterinary geriatrics (caring for old animals), the drug is used in dogs and cats over 7 years of age intramuscularly at a dose of 10 mg per 1 kg of animal weight 2 times a day for 5 to 7 days. In the next 20 – 25 days Mexidol-Vet is used in the form of tablets at a dose of 5 mg per 1 kg of animal weight. The repeated course is carried out in 3 – 4 months. The dose, duration of the course of treatment and the frequency of application of Mexidol-Vet are prescribed by the attending veterinarian, depending on the physiological state of the animal and the course of the disease. The use of Mexidol-Vet solution for injection does not exclude the use of other means of pathogenetic and symptomatic therapy.
SIDE EFFECTS
As a rule, there are no side effects and complications when using the drug in accordance with the instructions.
CONTRAINDICATIONS
Increased individual sensitivity to the drug.
SPECIAL INSTRUCTIONS
No special precautions are taken.
STORAGE CONDITIONS
With caution. Store in a dry, dark place and out of reach of children and animals at temperatures from 2 to 25 ºС. The shelf life is 3 years.
GENERAL INFORMATION
1. MEXIDOL-VET solution for injection (Solutio Mexidol-Vet pro injectionibus) and MEXIDOL-VET tablets (Tabulettae Mexidol-Vet) – a drug for the treatment and prevention of diseases in animals.
2. MEXIDOL-VET solution for injection as an active ingredient in 1 ml contains 25 mg or 50 mg of mexidol (ethylmethylhydroxypyridine succinate), as well as water for injection. Tablets contain 50 mg and 125 mg each. of the same active substance, as well as auxiliary form-forming components: lactose, potato starch, magnesium or calcium stearate.
3. Store MEXIDOL-VET in a dry, dark place at a temperature of 5 ° to 25 ° C.
The shelf life of the drug, subject to storage conditions, is 3 years from the date of manufacture. MEXIDOL-VET should not be used after the expiration date.
II. PHARMACOLOGICAL PROPERTIES
4. MEXIDOL-VET has pronounced antioxidant, antihypoxic and membrane-protective properties, increases the body’s resistance to the effects of various damaging factors to oxygen-dependent pathological conditions (shock, hypoxia and ischemia). The drug improves the metabolism of brain tissues and their blood supply. It stabilizes the membrane structures of blood cells (erythrocytes and platelets). It improves microcirculation and rheological properties of blood, reduces platelet aggregation, has stress-protective, anticonvulsant, tranquilizing and nootropic effects. It has a hypolipidemic effect, reduces the content of total cholesterol and low-density lipoproteins. MEXIDOL-VET inhibits lipid peroxidation, increases the activity of superoxide dismutase, modulates the activity of enzymes and membrane receptor complexes, improves synaptic transmission and transport of mediators. The mechanism of action of the drug is the ability to cause an increase in compensatory activation of aerobic glycolysis and to reduce the degree of inhibition of oxidative processes in the Krebs cycle under hypoxic conditions with an increase in the content of ATP and creatine phosphate; activate the energy-synthesizing function of mitochondria. When administered parenterally, the drug quickly enters the blood, organs and tissues of the animal. It is excreted from the body in the urine, mainly in the glucuronoconjugated form. activate the energy-synthesizing function of mitochondria. When administered parenterally, the drug quickly enters the blood, organs and tissues of the animal. It is excreted in the urine, mainly in the glucuronoconjugated form. activate the energy-synthesizing function of mitochondria. When administered parenterally, the drug quickly enters the blood, organs and tissues of the animal. It is excreted in the urine, mainly in the glucuronoconjugated form.
5. MEXIDOL-VET, according to the degree of impact on the body, is classified as a low-hazard substance, does not have local irritating, cumulative, embryotoxic and teratogenic properties.
III. ORDER OF APPLICATION OF THE PREPARATION
6. MEXIDOL-VET is prescribed to animals for therapeutic and prophylactic purposes independently and in complex therapy for the following indications: – Acute and chronic cardiovascular and cardiopulmonary insufficiency;
– Exogenous – organic diseases of the brain (as a result of neuroinfections), acute disorders of cerebral circulation, epilepsy, epileptic syndrome, traumatic brain injury;
– Prevention of anesthetic, post-anesthetic complications. Postoperative, post-traumatic period;
– Acute purulent – inflammatory processes of the abdominal cavity (acute necrotizing pancreatitis, peritonitis)
– Pathologies of the skin (dermatitis, eczema, trophic ulcers), prevention of complications in wound healing, improvement of the skin and hair;
– Periods of extreme loads, increased resistance under stresses associated with adaptation of animals to new conditions (transportation, participation in exhibitions, competitions);
– In veterinary geriatrics with age-related disorders of body functions (sluggish chronic diseases, neurodermatitis, dermatitis associated with impaired peripheral circulation ).
Method of administration and dosage :
7. MEXIDOL-VET solution for injection is administered to animals intramuscularly or intravenously. When administered intravenously, the drug is diluted with water for injection or saline. The therapeutic dose of the drug depends on the severity of the clinical condition of the animal. In the acute period of the disease, the drug is used starting from a dose of 10-15 mg / kg, then it is reduced to 5 mg / kg of animal weight. The course of treatment is 5-15 days. The daily dose of MEXIDOL-VET solution for injection is administered in two doses. In order to prevent anesthetic and post-anesthetic complications, the drug is used 2-3 days before the operation, in the postoperative period – within 2-3 days after the operation twice a day. With prolonged anesthesia, severe clinical condition of the animal, MEXIDOL-VET is administered during the operation in fractional doses up to 20 mg / kg of animal weight. As a therapeutic and prophylactic agent in veterinary geriatrics, MEXIDOL-VET solution for injection is used intramuscularly to animals over 7 years old at a dose of 10 mg / kg twice a day for 3-4 days. In the next 20-25 days, MEXIDOL-VET is used in the form of tablets at a dosage of 2.5-5.0 mg / kg. The repeated course is carried out in 3-4 months.
8. The dose, duration of the course and the frequency of application of MEXIDOL-VET are prescribed by the attending veterinarian, depending on the physiological state of the animal and the course of the disease.
9. The use of the drug MEXIDOL-VET does not exclude the use of other means of pathogenetic and symptomatic therapy.
10. Side effects and complications when using the drug in accordance with this instruction, as a rule, are not observed. Contraindications: individual intolerance to the components of the drug.
IV. PERSONAL PREVENTION MEASURES
11. When working with MEXIDOL-VET solution for injection, you should follow the general rules of personal hygiene and safety measures provided for when working with medicinal products.
12. MEXIDOL-VET solution for injection should be kept out of the reach of children.
The instruction was developed by OOO NPK Farmasoft, Moscow.
DOSAGE TABLE
Injection form
| Type and weight of the animal | Daily dose of MEXIDOL-VET solution for injection (per animal) | ||
| DV *, mg | 2.5% solution, ml | 5% solution, ml | |
| Cats | 25-50 | 1-2 | 0.5-1 |
| Small breed dogs | 50-100 | 2-4 | 1-2 |
| Medium breed dogs | 100-250 | → | 4-8 |
| Large breed dogs | 250-500 | → | 8-10 |
Tablet form
| Kind of animal | Therapeutic dose | Prophylactic dose | ||
| Chronic period of the disease | ||||
| Tablet | Tablet | DV 50 mg tablet | DV 125 mg tablet | |
| DV 50 mg | DV 125 mg | |||
| Cats, dogs of small breeds | 1 tab / 2 times a day | 1/3 tab / 2 times a day | 1/2 – 1 tablet / 1-2 times a day | 1/6 tab / 1-2 times a day |
| Medium breed dogs | 2-3 tablets / 2 times a day | 1 tab / 2 times a day | 1-2 tablets / 1-2 times a day | 1/2 tab / 1-2 times a day |
| Large breed dogs | 4-6 tablets / 2 times a day | 2 tablets / 2 times a day | 3 tablets / 1 – 2 times a day | 1 tablet / 1-2 times a day |