Online Pet Vet Pharmacy: Best Deals Up to 40% Off + FREE Shipping on Orders €100+

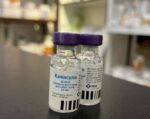


Caninsulin 2.5 ml Solution
40.10 € Original price was: 40.10 €.31.79 €Current price is: 31.79 €.
| Animals | Dogs |
|---|---|
| Brands | Msd, Usa |
| Form | Suspension/Emulsion/Syrup |
Caninsulin 2.5 ml (40 units/ml)
For the treatment of dogs and cats suffering from diabetes.
Description
Suspension for injections of white color.
Storage
1 ml of the drug contains the active substance:
- insulin – 40 IU.
Excipients: methyl parahydroxybenzoate; zinc chloride; sodium acetate trihydrate; sodium chloride; sodium hydroxide, water for injections.
Pharmacological properties
ATC vet classification code QА10AC03 – antidiabetic drugs.
Kaninsulin is a medium-acting drug containing amorphous and crystalline insulin in a ratio of 3.5:6.5.
Insulin is a hormone that ensures the transport of glucose into cells and activates intracellular enzymes that are responsible for the use and storage of glucose, amino acids and fatty acids. Insulin also suppresses the processes of catabolism, in particular, proteolysis, gluconeogenesis and lipolysis.
Diabetes mellitus is a disease characterized by an absolute or partial deficiency of insulin, which leads to persistent hyperglycemia. Accordingly, monitoring the concentration of glucose in the blood serum makes it possible to assess the systemic effect after the use of insulin.
In dogs with diabetes, the effect of Kaninsulin on the concentration of glucose in the blood reaches a maximum 6-8 hours after the injection and lasts up to 14-24 hours. In cats, the effect of Kaninsulin on the concentration of glucose in the blood reaches a maximum 4-6 hours after the injection and lasts up to 8-12 hours.
Kaninsulin is a medium-acting drug containing amorphous and crystalline insulin in a ratio of 3.5:6.5. In diabetic dogs, the peak concentration of insulin in blood plasma is observed 2-6 hours after subcutaneous injection, remaining at a level exceeding the pre-injection level for 14-24 hours. In cats, the peak concentration of insulin on the level of glucose in the blood plasma is observed 1.5 hours after subcutaneous injection, remaining at a level exceeding the pre-injection level for 5-12 hours.
Application
For the treatment of dogs and cats suffering from diabetes.
Dosage
The drug is injected 1 or 2 times a day, subcutaneously, choosing a different injection site each time using a 40-unit insulin syringe (from a vial) or a VetPen injector (from a cartridge).
The bottle is well shaken by rolling it between the palms until a homogeneous milky content is obtained.
Stabilization phase
Dogs
Therapy is started with an initial dose of 0.5 IU/kg of body weight per day.
| Body weight of the animal (kg) | Initial dose per animal (IU per day) |
| 5 | 2 |
| 10 | 5 |
| 15 | 7 |
| 20 | 10 |
Depending on the development of clinical signs and the results of blood glucose monitoring, the dose may be adjusted by 10%, but not more often than once every 3-7 days.
For some dogs, a two-time injection of the drug is necessary, in addition, after each injection of the drug, the dose is reduced by 25% of the calculated one. Thus, the total daily dose of the drug after a two-time injection will be less than double. For example, if a dog weighing 10 kg is calculated to need 5 IU of insulin per day, a two-time administration of 3 IU will be 25% less than 5 IU. The interval between injections is 12 hours. In the future, dose correction is carried out according to the same scheme.
Synchronize the feeding of the animal and the introduction of the drug, daily maintaining a constant composition of the diet and the regularity of feeding.
In dogs to which the drug is administered once a day, the second feeding usually coincides with the maximum effect of the drug. For a two-time administration, feeding practically coincides with the administration of the drug.
cats
The drug is initially administered in a dose of 1-2 IU, depending on the concentration of glucose in the blood. Cats are given the drug twice a day.
| Glucose concentration in blood serum | Initial dose for a cat |
| < 20 mmol/L or < 3.6 g/L (< 360 mg/dL) | 1 IU twice a day |
| ≥ 20 mmol/L or ≥ 3.6 g/L (≥ 360 mg/dL) | 2 IU twice a day |
The composition of the diet and the regularity of feeding do not change.
Correction of the daily dose of the drug is carried out no more than once a week, depending on the results of measuring the level of glucose in the blood serum. The dose increase should not exceed 1 IU. Ideally, no more than 2 IU per day should be administered during the first three weeks of treatment.
*Kilograms to Pounds conversion table
| Kilograms (kg) | Pounds (lb) | Pounds+Ounces (lb+oz) |
| 0.1 kg | 0.220 lb | 0 lb 3.527 oz |
| 1 kg | 2.205 lb | 2 lb 3.274 oz |
| 5 kg | 11.023 lb | 11 lb 0.370 oz |
| 10 kg | 22.046 lb | 22 lb 0.740 oz |
Support phase
After establishing the effective effect of the drug in a maintenance dose, a long-term treatment program is started with mandatory regular monitoring of the blood glucose level.
Subsequent re-examinations are carried out after 2-4 months (or more often). The dose of the drug is adjusted based on the results of the blood test and the animal’s clinical condition.
Pet owners should be thoroughly informed about the clinical signs of hypo-/hyperglycemia.
Contraindication
Do not administer the drug intravenously.
Do not use in animals in a ketoacidotic state.
Do not inject a drug that contains clots that do not break after shaking the vial.
Side effect
Very rarely, an allergic reaction to the introduction of foreign insulin is possible.
Special precautions for use
The drug is used in animals with a regular feeding schedule.
Owners of animals to whom treatment with the drug is prescribed should be instructed about the need to have powdered sugar or honey at home. Animals with clinical symptoms of hypoglycemia: hunger, locomotion, muscle spasms, stumbling of the hind limbs and disorientation need urgent administration of glucose solution or feeding with honey (powdered sugar) in order to restore the concentration of glucose in the blood.
Interaction with other means or other forms of interaction
Do not enter together with progestagens. Simultaneous use with corticosteroids can affect the effectiveness of the drug, so dose correction is necessary.
Special warnings
The drug is administered using a 40 U insulin syringe (from a bottle) or a VetPen injector (from a cartridge).
Special precautions for persons and service personnel who administer the drug
The drug is administered by a doctor of veterinary medicine. In case of contact of the drug with human skin, it is necessary to thoroughly wash the place of contact with soap and water. After contact with the eyes and mucous membranes, rinse with plenty of warm water.
In case of accidental administration of the drug to a person, it is necessary to immediately consult a doctor, showing him the package or leaflet-tab.
Release form
Vials made of type I glass (according to the European Pharmacopoeia), closed with rubber stoppers under an aluminum run-in of 2.5 ml. Secondary packaging – cardboard boxes of 1 or 10 vials.
Vials made of type I glass (according to the European Pharmacopoeia), closed with rubber stoppers under an aluminum run-in, 10 ml each. Secondary packaging – cardboard boxes.
Cartridges made of type I glass (according to the European Pharmacopoeia) with a piston, closed with rubber caps under an aluminum break-in of 2.7 ml. Secondary packaging – cardboard boxes of 10 pcs.
Storage
Shelf life – 24 months.
After the first selection from the bottle – 42 days.
After the first selection from the cartridge – 28 days.
Store in a dry, dark place out of the reach of children at a temperature of 2° to 8°C. After the first selection, vials or cartridges with the drug are stored at temperatures from 2° to 25°C.
For use in veterinary medicine!